Our research
Myo-inositol phosphates, soluble and lipid molecules, play fundamental functions in cell regulation and membrane dynamics. Phosphatidylinositol, a membrane phospholipid, can be reversibly phosphorylated at the 3, 4 and 5 positions of myo-inositol to generate phosphoinositides (PIs). Seven PIs are known in mammalian cells. PIs play fundamental roles in cell physiology, signaling and physiopathology (Blero et al., 2007). Moreover, the phosphoinositide (PI) 3-kinase (PI3K) pathway is one of the most frequently mutated signaling pathways in cancer being actively pursued today as a therapeutic target. However, resistance to inhibitors to PI3K due to compensatory signaling and genomic changes have emerged, suggesting the need for the use of inhibitors in combination with novel strategies and targets. The role of phosphatidylinositol 3,4,5-trisphosphate (PI(3,4,5)P3), the product of PI3K is fundamental in cancer research and often considered as “oncogenic” but other PIs such as phosphatidylinositol 3,4-bisphosphate (PI(3,4)P2) and phosphatidylinositol 4,5-bisphosphate (PI(4,5)P2) are also critical signal molecules, particularly in the control of cell adhesion, migration or invasion.
Our interest in the PI metabolism started with the cloning of a series of inositol polyphosphate 5-phosphatase and Ins(1,4,5)P3 3-kinases (the enzymes that phosphorylate Ins(1,4,5)P3 to Ins(1,3,4,5)P4).
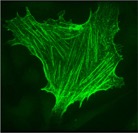
Inositol trisphosphate 3-kinase A staining (in green)
cDNAs encoding INPP5A, Itpka, b and c followed by the SH2 domain containing inositol 5-phosphatases SHIP1 and SHIP2 have been cloned. The substrate of SHIP1/2 is PI(3,4,5)P3 and the product of the reaction is PI(3,4)P2. Both PIs are able to bind to PKB and PDK1 PH domains and to facilitate PDK1 phosphorylation of PKB in vitro.
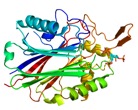 SHIP2 active site
SHIP2 active site
SHIP1/2 are very much studied due to their implication in immune response, myeloid cell survival for SHIP1 and developmental aspects for SHIP2 (Clement et al., 2001; Huber et al., 1999; Dubois et al., 2012). In addition, SHIP1/2 implication in human cancer has been strongly suggested next to the established tumor suppressor role of PTEN (Erneux et al., 2011).
The current goal of our laboratory is to define the oncogenic and anti-oncogenic function of PI 5-phosphatases in cancer cells particularly in glioblastoma and breast cancer. The way these enzymes are regulated and/or participate in non-enzymatic properties but rather through docking properties is another aspect of our research.
Group members
Contact:

Christophe Erneux,
phone # 32 2 555 4162 (4161).
Current lab members:
Somadri Ghosh (Postdoc), Ana Raquel Ramos (Postdoc)
Alumni:
Dominique Couchie, Françoise Miot, Kazunaga Takazawa (Japan), Anne Delvaux, Benoît Verjans, Valérie Vanweyenberg, David Communi, Florence De Smedt, Xavier Pesesse, Valérie Dewaste, Nathalie Paternotte, Sylvie Giuriato (France), Lyndsay A Drayer (Holland), Daniel Blero, Katrien Backers, Jing Zhang (China), Fabrice Vandeput, Alexandre Leyman, Laurence Deneubourg, Sandra Koenig.
Publications
Publications (full list)
Major Publications
1. Erneux C. S Cohen, D L Garbers (1983) The kinetics of tyrosine phosphorylation by the purified epidermal growth factor receptor kinase of A-431 cells. J Biol Chem 258: 7. 4137-4142
2. Takazawa K., Vandekerckhove J., Dumont J.E. and Erneux C. (1990) Cloning and expression in Escherichia coli of a rat brain cDNA encoding a Ca2+/calmodulin-sensitive inol 1,4,5-trisphosphate 3-kinase. Biochem.J. 272: 107-112.
3. Verjans B., Desmedt F., Lecocq R., Vanweyenberg V., Moreau C., Erneux C. Cloning and expression in Escherichia coli of a dog thyroid cDNA encoding an inositol 1,4,5-trisphosphate 5-phosphatase. Biochem. J. 1994 300: 85-90.
4. Communi, D., Vanweyenberg, V., Erneux, C. D-myo-inositol 1,4,5-trisphosphate 3-kinase-A is activated by receptor activation by a calcium:calmodulin-dependent protein kinase II phosphorylation mechanism EMBO J. 1997 16: 1943-1952.
5. Pesesse, X., Deleu, S., De Smedt, F., Drayer L.A. , Erneux C. (1997) Identification of a second SH2-domain-containing protein closely related to the phosphatidylinositol polyphosphate 5-phosphatase SHIP. Biochem Biophys Res Commun 239: 3. 697-700.
6. De Smedt, F., Missiaen, L., Parys, J. B., Vanweyenberg, De Smedt, J, Erneux, C. Isoprenylated human brain type I inositol 1,4,5-trisphosphate 5-phosphatase controls Ca2+ oscillations induced by ATP in Chinese Hamster Ovary cells J. Biol. Chem. 1997 272: 17367-17375.
7. Vandeput F., Combettes L., Mills, S.J., Backers, K., Wohlkonig, A., Parys, J.B., De Smedt, H., Missiaen, L., Dupont, G., Potter, B.V.L. and Erneux, C. Biphenyl 2,3′,4,5′,6-pentakisphosphate, a novel inositol polyphosphate surrogate, modulates Ca2+ responses in rat hepatocytes FASEB J 2007 21, 1481-1491.
8. Elong Edimo W, Derua R, Janssens V, Nakamura T, Vanderwinden JM, Waelkens E, and Erneux C. Evidence of SHIP2 S132 phosphorylation, its nuclear localization and stability. Biochem J 2011 439(3):391-401.
9. Edimo WE, Ghosh S, Derua R, Janssens V, Waelkens E, Vanderwinden JM, Robe P, and Erneux C . SHIP2 controls plasma membrane PI(4,5)P2 thereby participating in the control of cell migration in 1321 N1 glioblastoma. J Cell Sci 2016 129, 1101-1114.
10. Vandenberghe P, Hagué P, Hockman SC, Manganiello VC, Demetter P, Erneux C*, Vanderwinden JM*. Phosphodiesterase 3A: a new player in development of interstitial cells of Cajal and a prospective target in gastrointestinal stromal tumors (GIST). Oncotarget. 2017 Jun 20;8(25):41026-41043. *equal contribution of last two authors
11. Inhibition of SHIP2 activity inhibits cell migration and could prevent metastasis in breast cancer cells”, Somadri Ghosh, Samuel Scozzaro, Ana Raquel Ramos, Sebastien Delcambre, Clement Chevalier, Pavel Krejci, and Christophe Erneux. J Cell Sci. 2018 Aug 16;131(16).
12. The impact of phosphoinositide 5-phosphatases on phosphoinositides in cell function and human disease. Ramos AR, Ghosh S, Erneux C. J Lipid Res. 2019 Feb;60(2):276-286.
13. Lipid phosphatases SKIP and SHIP2 regulate fibronectin-dependent cell migration in glioblastoma. Ramos AR, Ghosh S, Dedobbeleer M, Robe PA, Rogister B, Erneux C. FEBS J. 2019 Jan 29.