BRIEF OVERVIEW ONGOING RESEARCH CDK4 and cancer. As the master integrator of G1 phase cell cycle regulations, CDK4 is the first CDK to be activated in response to mitogenic stimuli and all their oncogenic perversions. Its main function is the inactivation of the pRb antioncogene (Rb1 gene). CDK4 activity requires its binding to a cyclin D (CCND1-3 genes) competed by INK4 CDK4 inhibitors such as p16 (CDKN2A-D). Genetic ablation of both CDK4 and CDK6 or of the three cyclins D permits normal cell cycles involved in development (due to cell cycle regulation plasticity and compensation by other CDKs (3)) but its deregulation causes addiction to CDK4 activity. Indeed, in different cancer models including breast cancers and NSC lung cancers, acute inhibition of CDK4/6 by the specific PD0332991 compound (Palbociclib) specifically induces either senescence or apoptotic cell death of tumoral cells. This and other CDK4/6 inhibitory drugs developed by Novartis (ribocliclib) or Eli Lilly (abemaciclib) are tested in a growing number of phase II/III clinical trials against various pRb-proficient chemotherapy-resistant cancers (90 studies with a total of 16000 patients are recorded in ClinicalTrials.gov). Despite mild dose-limiting side effects (cytopenia,..), palbociclib and ribociclib induce an ‘unprecedented improvement of progression-free survival’ of women with advanced ER-positive breast cancers, leading to the approval of palbociclib (FDA, Feb. 2015; EMA, Nov. 2016) and ribociclib (FDA, March 2017) as first-line treatments of advanced ER-positive breast cancer combined with endocrine therapy. Extension of CDK4/6 inhibitors to other cancers is limited by the lack of suitable biomarker of potential sensitivity.
phone # +32(0)2 555 4153 Eric Raspé, PhD (eraspe@ulb.ac.be) Eric received a PhD in Chemistry from ULB (IRIBHM) in 1991. He was then « chargé de recherches » of the FNRS at the IRIBHM. From 1995 till 2004 he was a research associate at the Institut Pasteur of Lille and then Genfit (Lille) in charge of Lipha/Merck Santé research programs related to diabetes and atherosis. In 2004 he joined Geert Berx’s lab at the VIB (Ghent University) as a senior research associate (involvement of EMT in breast cancer) and he acquired a strong expertise in bioinformatics and breast cancers. He came back to our lab in 2011 within the framework of a Welbio program. Katia received a PhD in Biomedical Sciences from ULB (IRIBHM) in 2000. While pursuing her research activities in our lab, she was then appointed as a teaching assistant in the Chemistry Department of the ULB Medical School till 2008. From then she was pursuing her research as a senior research assistant supported by the Télévie (FNRS) and then Welbio. Sabine performed her thesis research work in our lab and received a PhD in Biomedical Sciences in 2006. She was « chargé de recherches » of the FNRS till 2010 and received a Télévie/FNRS postdoctoral grant. As a senior research assistant, she is now supported by the FNRS. Jaime received his PhD from the Unversity of Lisbon in 2013. He worked on the molecular characterization of dedifferentiated thyroid cancers. He joined our lab in 2013 thanks to a Televie fellowship. Bachelor in Clinical Chemistry 1981; Industrial Draftman Diploma 1989 PhD Theses: Colleoni Bianca (2017) : Critical mechanisms of CDK4 activation, the key of cell cycle commitment and an essential target of oncogenic processes. Roles of p21 phosphorylations and identification of novel CDK4 activating kinases. (Adviser : P. Roger) Bisteau Xavier (2013): Activation de la CDK4, clé de l’engagement du cycle cellulaire et carrefour des voies oncogéniques. Evaluation de l’implication de la kinase activatrice des CDKs (CAK) et des phosphorylations de p21. (Adviser : P. Roger). Xavier is now in Philipp Kaldis cell cycle lab in Singapore. (xbisteau@gmail.com) Blancquaert Sara (2010) : cAMP-dependent signal transduction leading to mitogenesis in thyroid. Implication of the mammalian target of rapamycin (mTOR). (Adviser : P. Roger) Arsenijevic Tatjana (2006): L’identification d’une nouvelle protéine partenaire de la cycline D3, l’AKAP95. (Advisers : I. Pirson, P. Roger) Bockstaele Laurence (2006): Réévaluation de la régulation de l’activité de la CDK4 (kinase dépendante des cyclines D), clé de l’engagement dans le cycle cellulaire. Rôle de l'”inhibiteur” p27Kip1. (Adviser : P. Roger) Paternot Sabine (2006): Différents mécanismes d’activation de la CDK4 par l’AMP cyclique et les facteurs de croissance dans les cellules épithéliales thyroïdiennes. (Adviser : P. Roger) Coulonval Katia (2000) : Etude des voies de signalisation conduisant à la mitogénèse dans la cellule thyroïdienne. (Advisers : J.E. Dumont, F. Lamy) Van Keymeulen Alexandra (2000) : Régulation synergique de la mitogenèse par l’AMPc et des facteurs permissifs dans la cellule thyroïdienne. (Adviser : P. Roger) Depoortere Fabienne (1999) : Régulation de la phase G1 du cycle cellulaire du thyrocyte par l’AMPc et le TGF. (Adviser : P. Roger) Baptist Mireille (1995) : Nouveaux aspects de la régulation du cycle de division des cellules épithéliales thyroïdiennes par l’AMPc. (Adviser : P. Roger) Raspé Eric (1991) : Etude du contrôle et du rôle physiologique de la cascade du Ca2+- phosphatidylinositol du thyrocyte. (Adviser : J.E. Dumont)
Publications (Full) http://publicationslist.org/pierre.roger Publications 2023 Anaplastic thyroid carcinoma: advances in molecular profiling and targeted therapy. Jungels C, Pita JM, Costante G. Curr Opin Oncol. 2023 Jan 1;35(1):1-9. doi: 10.1097/CCO.0000000000000918. Epub 2022 Nov 18. CDK4/6 Inhibitors in Pancreatobiliary Cancers: Opportunities and Challenges. Arsenijevic T, Coulonval K, Raspé E, Demols A, Roger PP, Van Laethem JL. Cancers (Basel). 2023 Feb 3;15(3):968. doi: 10.3390/cancers15030968. RHOJ controls EMT-associated resistance to chemotherapy. Debaugnies M, Rodríguez-Acebes S, Blondeau J, Parent MA, Zocco M, Song Y, de Maertelaer V, Moers V, Latil M, Dubois C, Coulonval K, Impens F, Van Haver D, Dufour S, Uemura A, Sotiropoulou PA, Méndez J, Blanpain C. Nature. 2023 Apr;616(7955):168-175. doi: 10.1038/s41586-023-05838-7. Epub 2023 Mar 22. CDK4 phosphorylation status and rational use for combining CDK4/6 and BRAF/MEK inhibition in advanced thyroid carcinomas. Pita JM, Raspé E, Coulonval K, Decaussin-Petrucci M, Tarabichi M, Dom G, Libert F, Craciun L, Andry G, Wicquart L, Leteurtre E, Trésallet C, Marlow LA, Copland JA, Durante C, Maenhaut C, Cavaco BM, Dumont JE, Costante G, Roger PP. Front Endocrinol (Lausanne). 2023 Oct 26;14:1247542. doi: 10.3389/fendo.2023.1247542. 50 Main publications ROGER, P.P., HOTIMSKY, A., MOREAU, C., DUMONT, J.E.
Stimulation by thyrotropin, choleratoxin and dibutyryl cyclic AMP of the multiplication of differentiated thyroid cells in vitro.
Mol. Cell. Endocrinol. 26, 165-176 (1982). ROGER, P.P., SERVAIS, P., DUMONT, J.E.
Stimulation by thyrotropin and cyclic AMP of the proliferation of quiescent canine thyroid cells cultured in a defined medium containing insulin.
FEBS Lett. 157, 323-329 (1983). ROGER, P.P., DUMONT, J.E.
Factors controlling proliferation and differentiation of canine thyroid cells cultured in reduced serum conditions: effects of thyrotropin, cyclic AMP and growth factors.
Mol. Cell. Endocrinol. 36, 79-93 (1984). ROGER, P.P., VAN HEUVERSWYN, B., LAMBERT, C., REUSE, S., VASSART, G., DUMONT, J.E.
Antagonistic effects of thyrotropin and epidermal growth factor on thyroglobulin mRNA level in cultured thyroid cells.
Eur. J. Biochem. 152, 239-245 (1985). ROGER, P.P., REUSE, S., SERVAIS, P., VAN HEUVERSWYN, B., DUMONT, J.E.
Stimulation of cell proliferation and inhibition of differentiation expression by tumor-promoting phorbol esters in dog thyroid cells in primary culture.
Cancer Res. 46, 898-906 (1986). ROGER, P.P., SERVAIS, P., DUMONT, J.E.
Induction of DNA synthesis in dog thyrocytes in primary culture: synergistic effects of thyrotropin and cyclic AMP with epidermal growth factor and insulin.
J. Cell. Physiol. 130, 58-67 (1987). ROGER, P.P., SERVAIS, P., DUMONT, J.E.
Regulation of dog thyroid epithelial cell cycle by forskolin, an adenylate cyclase activator.
Exp. Cell Res. 172, 282-292 (1987). DUMONT, J.E., ROGER, P.P., LUDGATE, M.
Assays for thyroid growth immunoglobulins and their clinical applications : methods, concepts and misconceptions.
Endocrine Rev. 8, 448-452 (1987). ROGER, P.P., TATON, M., VAN SANDE, J., DUMONT, J.E.
Mitogenic effects of thyrotropin and cyclic AMP in differentiated human thyroid cells in vitro.
J. Clin. Endocrinol. Metab. 66, 1158-1165 (1988). DUMONT, J.E., JAUNIAUX, J.C., ROGER, P.P.
The cyclic AMP-mediated stimulation of cell proliferation.
Trends Biochem. Sci. 14, 67-71, (1989). ROGER, P.P., RICKAERT, F., LAMY, F., AUTHELET, M., DUMONT, J.E.
Actin stress fiber disruption and tropomyosin isoform switching in normal thyroid epithelial cells stimulated by thyrotropin and phorbol esters.
Exp. Cell. Res. 182, 1-13 (1989). POHL, V., ROGER, P.P., CHRISTOPHE, D., PATTYN, G., VASSART, G., DUMONT, J.E.
Differentiation expression during proliferative activity induced through different pathways: in situ hybridization study of thyroglobulin gene expression in thyroid epithelial cells.
J. Cell Biol. 111, 663-672 (1990). LAMY, F., TATON, M., DUMONT, J.E., ROGER, P.P.
Control of protein synthesis by thyrotropin and epidermal growth factor in human thyrocytes : role of morphological changes.
Mol. Cell. Endocrinol. 73, 195-209 (1990). COCLET, J., LAMY, F., RICKAERT, F., DUMONT, J.E., ROGER, P.P.
Intermediate filaments in normal thyrocytes : modulation of vimentin expression in primary cultures.
Mol. Cell Endocrinol. 76, 135-148 (1991). DUMONT, J.E., MAENHAUT, C., PIRSON, I., BAPTIST, M., ROGER, P.P.
Growth factors controlling the thyroid gland. ROGER, P.P., BAPTIST, M., DUMONT, J.E.
A mechanism generating heterogeneity in thyroid epithelial cells : suppression of the thyrotropin/cAMP-dependent mitogenic pathway after cell division induced by cAMP-independent factors.
J. Cell Biol. 117, 383-393 (1992). DUMONT, J.E., LAMY, F., ROGER, P., MAENHAUT, C.
Physiological and pathological regulation of thyroid cell proliferation and differentiation by thyrotropin and other factors: an example of positive control of proliferation and differentiation by the cyclic AMP cascade.
Physiol. Rev. 72, 667-697 (1992). BAPTIST, M., DUMONT, J.E., ROGER, P.P.
Demonstration of cell cycle kinetics in thyroid primary culture by immunostaining of proliferating cell nuclear antigen : differences in cyclic AMP-dependent and independent mitogenic stimulations.
J. Cell. Sci. 105, 69-80 (1993). ROGER, P.P., REUSE, S., MAENHAUT, C., DUMONT, J.E.
Multiple facets of the modulation of growth by cyclic AMP BAPTIST, M., DUMONT, J.E., ROGER, P.P.
Intercellular heterogeneity of early mitogenic events: cAMP generalizes the EGF effect on c-Fos protein appearance but not on MAP kinase phosphorylation and nuclear translocation in dog thyroid epithelial cells.
Exp. Cell Res. 221, 160-171 (1995). BAPTIST, M., LAMY, F., GANNON, J., HUNT, T., DUMONT, J.E., ROGER, P.P.
Expression and subcellular localization of CDK2 and cdc2 kinases and their common partner cyclin A in thyroid epithelial cells: comparison of cyclic AMP-dependent and -independent cell cycles.
J. Cell. Physiol. 166, 256-273 (1996). DEPOORTERE, F., DUMONT, J.E., ROGER, P.P.
Paradoxical accumulation of the cyclin-dependent kinase inhibitor p27KIP1 during the cAMP-dependent mitogenic stimulation of thyroid epithelial cells.
J. Cell Sci., 109, 1759-1764 (1996). BURIKHANOV, R., COULONVAL, K., PIRSON, I., LAMY, F., DUMONT, J.E., ROGER, P.P.
Thyrotropin via cyclic AMP induces insulin receptor expression and insulin co-stimulation of growth and amplifies insulin and insulin-like growth factor signalling pathways in dog thyroid epithelial cells.
J. Biol. Chem. 271, 29400-29406 (1996). ROGER, P.P., CHRISTOPHE, D., DUMONT, J.E., PIRSON, I.
The dog thyroid primary culture system: a model of the regulation of function, growth and differentiation by cyclic AMP and other well defined signaling cascades (invited review).
Eur. J. Endocrinol. 137, 579-598 (1997). Depoortere, F., Van Keymeulen, A., Lukas, J., Costagliola, S., Bartkova, J., Dumont, J.E., Bartek, J., Roger, P.P. (corresponding author), Dremier, S.
A requirement for cyclin D3-cyclin-dependent kinase (cdk)-4 assembly in the cyclic adenosine monophosphate-dependent proliferation of thyrocytes.
J. Cell Biol. 140, 1427-1439 (1998). ROGER, P.P., DEMARTIN, S., DUMONT, J.E. VAN KEYMEULEN, A., BARTEK, J., DUMONT, J.E., ROGER, P.P. DEPOORTERE, F., PIRSON, I., BARTEK, J., DUMONT, P.P., ROGER, P.P. VAN KEYMEULEN, A., DELEU, S., BARTEK, J., DUMONT, J.E., ROGER, P.P KIMURA, T., VAN KEYMEULEN, A., GOLSTEIN, J., FUSCO, A., DUMONT, J.E., ROGER, P.P. PATERNOT, S., COULONVAL, K., DUMONT, J.E., ROGER, P.P. COULONVAL, K., BOCKSTAELE, L., PATERNOT, S., DUMONT, J.E., ROGER, P.P. COULONVAL, K., BOCKSTAELE, L., PATERNOT, S., ROGER, P.P. ARSENIJEVIC, T., DEGRAEF, C., DUMONT, J.E., ROGER, P.P., PIRSON, I. FORTEMAISON, F., BLANCQUAERT, S., DUMONT, J.E., MAENHAUT, C., AKTORIES, K., ROGER, P.P.(corresponding author), DREMIER, S. PATERNOT, S., ARSENIJEVIC, T., COULONVAL, K., BOCKSTAELE, L., DUMONT, J.E., ROGER, P.P. BOCKSTAELE, L., KOOKEN, H., LIBERT, F., PATERNOT, S., DUMONT, J.E., de LAUNOIT, Y., ROGER, P.P.(corresponding author), COULONVAL, K. PATERNOT, S., DUMONT, J.E., ROGER, P.P. BOCKSTAELE, L., COULONVAL, K., KOOKEN, H., PATERNOT, S., ROGER, P.P. DREMIER, S., MILENKOVIC, M., BLANCQUAERT, S., DUMONT, J.E., DOSKELAND, S.O., MAENHAUT, C., ROGER, P.P. ROCHA, A.S., PATERNOT, S., COULONVAL, K., DUMONT, J.E., SOARES, P., ROGER P.P. PATERNOT, S., ROGER, P.P. BOCKSTAELE, L.*, BISTEAU, X.* (*equal contribution), PATERNOT, S., ROGER, P.P. PATERNOT, S., BOCKSTAELE, L., BISTEAU, X., KOOKEN, H., COULONVAL, K., ROGER, P.P. BLANCQUAERT, S, WANG, L, PATERNOT, S, COULONVAL, K, DUMONT, JE, HARRIS, TE, ROGER, PP. COULONVAL, K., KOOKEN, H., ROGER, P.P. BISTEAU, X.*, PATERNOT, S.* (*equal contribution), COLLEONI, B., COULONVAL, K., ECKER, K., DE GROOTE, P., DECLERCQ, W., HENGST, L., ROGER, P.P. PATERNOT, S., COLLEONI, B., BISTEAU, X., ROGER, P.P. BIANCA COLLEONI*, SABINE PATERNOT* (equal contributions), JAIME M PITA, XAVIER BISTEAU, KATIA COULONVAL, ROGER J DAVIS, ERIC RASPÉ, PIERRE P. ROGER. ERIC RASPÉ, KATIA COULONVAL*, JAIME M. PITA*, SABINE PATERNOT*, FRANÇOISE ROTHÉ* (essential contributions), LAURE TWYFFELS, SYLVAIN BROHÉE, LIGIA CRACIUN, DENIS LARSIMONT, VÉRONIQUE KRUYS, FLAVIENNE SANDRAS, ISABELLE SALMON, STEVEN VAN LAERE, MARTINE PICCART, MICHAIL IGNATIADIS, CHRISTOS SOTIRIOU, PIERRE P. ROGER. SOME OTHER PUBLICATIONS BY MEMBERS OF THE GROUP COULONVAL K, VANDEPUT F, STEIN RC, KOZMA SC, LAMY F, DUMONT JE. RASPÉ E, DUEZ H, GERVOIS P, FIÉVET C, FRUCHART JC, BESNARD S, MARIANI J, TEDGUI A, BERX G, RASPÉ E, CHRISTOFORI G, THIERY JP, SLEEMAN JP. RASPE E, DECRAENE C, BERX G. FLOOR SL, DUMONT JE, MAENHAUT C, RASPE E. PITA JM, BANITO A, CAVACO BM, LEITE V. PITA JM, FIGUEIREDO IF, MOURA MM, LEITE V, CAVACO BM.Our research
Pierre Roger pioneered the study of proliferation and differentiation of thyroid cells by defining model systems of primary cultures in chemically defined medium (without serum). Using these systems, he demonstrated and characterized the unique coexistence in these cells of two distinct proliferation modes, activated by TSH (only via cAMP elevation and PKA activation) or by several growth factors (via Ras, MAP kinase and PI3kinase cascades). The cAMP pathway simultaneously stimulates proliferation and differentiation expression in thyrocytes, and is involved in goitrigenesis and generation of hyperfunctional adenomas, whereas the growth factor pathways induce dedifferentiation and are involved in thyroid carcinomas. In thyroid primary cultures, the positive cell cycle regulation by cAMP is unique as it targets the assembly and then the activation of complexes formed by preexisting cyclin D3 and cyclin-dependent kinase (CDK) 4, without involving most intermediaries of classical mitogenic signaling cascades.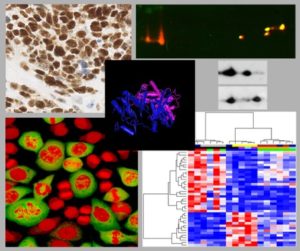
The present research interests of our group include the cellular and molecular features of cell cycle regulation in this model system and several others (investigation of cyclins, CDKs, their inhibitors, the interactions between these different proteins and their posttranslational modifications). Using two-dimensional electrophoresis to separate the phosphorylated forms of these proteins within their various complexes (see MORE..) we identified the activating phosphorylation of CDK4 as a direct crucial target for cell cycle regulation in various cell systems. Using various molecular, cellular, proteomic and bioinformatic approaches, we are currently (i) exploring the mechanisms of this critical CDK4 regulation that determines the inactivation of the central oncosuppressor protein pRb and the cell cycle decision in normal and cancerous cells; (ii) developing novel tools to predict whether cancer patients will benefit or not of treatments with the new CDK4 inhibitory drugs that are now approved for treatment of advanced ER+ breast cancers.Group members

Pierre P. Roger, PhD (PI) (proger@ulb.ac.be)

Katia Coulonval, PhD (kcoulonv@ulb.ac.be)
Sabine Paternot, PhD (spaterno@ulb.ac.be)
Jaime Pita, PhD(jaimpita@ulb.ac.be)
Vincent VercruyssePublications
Baillière’s Clinical Endocrinol. Metab. 5, 727-754 (1991).
Vitamins and Hormones, 51, 59-191 (1995).
Nature of the critical labile event that controls RB phosphorylation in the cyclic AMP-dependent cell cycle of thyrocytes in primary culture.
Exp. Cell Res. 252, 492-498 (1999).
Cyclin D3 accumulation and activity integrate and rank the comitogenic pathways of thyrotropin and insulin in thyrocytes in primary culture.
Oncogene 18, 7351-7359 (1999).
Transforming growth factor 1 selectively inhibits the cyclic AMP-dependent proliferation of primary thyroid epithelial cells by preventing the association of cyclin D3-cdk4 with nuclear p27kip1.
Mol. Biol. Cell 11, 1061-1076 (2000).
Respective roles of carbamylcholine and cyclic adenosine monophosphate in their synergistic regulation of cell cycle in thyroid primary cultures.
Endocrinology 142, 1251-1259 (2001).
Regulation of thyroid cell proliferation by TSH and other factors : a critical evaluation of in vitro models.
Endocrine Rev. 22, 631-656 (2001).
Cyclic AMP-dependent phosphorylation of cyclin D3-bound CDK4 determines the passage through the cell cycle restriction point in thyroid epithelial cells.
J. Biol. Chem. 278, 26533-26540 (2003).
The cyclin D3-CDK4-p27kip1 holoenzyme in thyroid epithelial cells : activation by TSH, inhibition by TGF, and phosphorylations of its subunits demonstrated by two-dimensional gel electrophoresis.
Exp. Cell Res. 291, 135-149 (2003).
Phosphorylations of cyclin-dependent kinase 2 revisited using two-dimensional gel electrophoresis.
J. Biol. Chem. 278, 52052-52060 (2003).
A novel partner for cyclins D: Protein Kinase A-anchoring protein AKAP95.
Biochem. J. 378, 673-679 (2004).
Differential involvement of the actin cytoskeleton in differentiation and mitogenesis of thyroid cells : Inactivation of Rho proteins contributes to cAMP-dependent gene expression but prevents mitogenesis.
Endocrinology 146, 5845-5495 (2005).
Distinct specificities of pRb phosphorylation by CDK4 activated by cyclin D1 or cyclin D3: differential involvement in the distinct mitogenic modes of thyroid epithelial cells.
Cell Cycle 5, 61-70 (2006).
Thr172-phosphorylation of CDK4: its relationship with cyclins and CDK “inhibitors”.
Mol. Cell Biol. 26, 5070-5085 (2006).
Differential utilization of cyclin D1 and cyclin D3 in the distinct mitogenic stimulations by growth factors and TSH of human thyrocytes in primary culture.
Mol. Endocrinol. 20, 3279-3292 (2006).
Regulation of CDK4 (invited review).
Cell Div. (BioMed Central) 1, n° 25 (2006).
http://www.celldiv.com/content/1/1/25.
Cyclic adenosine 3′,5′-monophosphate (cAMP)-dependent protein kinases, but not exchange proteins directly activated by cAMP (Epac), mediate thyrotropin/cAMP-dependent regulation of thyroid cells.
Endocrinology, 148, 4612-4622 (2007).
Cyclic AMP inhibits the proliferation of thyroid carcinoma cell lines through regulation of CDK4 phosphorylation.
Mol. Biol. Cell, 19, 4814-4825 (2008).
Combined inhibition of MEK and mammalian target of rapamycin abolishes phosphorylation of cyclin-dependent kinase 4 in glioblastoma cell lines and prevents their proliferation.
Cancer Res. 69, 4577-4581 (2009).
Differential regulation of CDK4 and CDK6, evidence that CDK4 might not be activated by CDK7, and design of a CDK6 activating mutation.
Mol. Cell. Biol., 29,4188-4200 (2009).
Rb inactivation in cell cycle and cancer : The puzzle of highly regulated activating phosphorylation of CDK4 versus constitutively active CDK-activating kinase (invited perspective).
Cell Cycle 9, 689-699 (2010).
Cyclic AMP-dependent activation of mammalian target of rapamycin (mTOR) in thyroid cells. Implication in mitogenesis and activation of CDK4.
Mol. Endocrinol. 24, 1453-1468 (2010).
Coupling of T161 and T14 phosphorylations protects cyclin B-CDK1 from premature activation.
Mol. Biol. Cell, 22, 3971-3985 (2011).
CDK4 T172-phosphorylation is central in a CDK7-dependent bidirectional CDK4/CDK2 interplay mediated by p21 phosphorylation at the restriction point.
PLoS Genetics, 9(5) e1003546 (2013)(21 pages ; 11 suppl. figures).
The CDK4/CDK6 inhibitor PD0332991 paradoxically stabilizes activated cyclin D3-CDK4/6 complexes.
Cell Cycle, 13, 2879-2888 (2014).
JNKs function as CDK4-activating kinases by phosphorylating CDK4 and p21.
Oncogene 36(30):4349-4361 (2017). doi: 10.1038/onc.2017.7.
CDK4 phosphorylation status and corresponding gene expression profile predict sensitivity to Palbociclib.
EMBO Mol. Med. 9(8):1052-1066 (2017). doi: 10.15252/emmm.201607084.
Phosphatidylinositol 3-kinase, protein kinase B and ribosomal S6 kinases in the stimulation of thyroid epithelial cell proliferation by cAMP and growth factors in the presence of insulin.
Biochem J. 348, 351-8 (2000).
STAELS B.
Transcriptional regulation of apolipoprotein C-III gene expression by the orphan nuclear receptor RORalpha.
J Biol Chem. 276, 2865-71 (2001).
Pre-EMTing metastasis? Recapitulation of morphogenetic processes in cancer.
Clin Exp Metastasis. 24, 587-97 (2007).
Gene expression profiling to dissect the complexity of cancer biology: pitfalls and promise.
Semin Cancer Biol. 22, 250-60 (2012).
Hallmarks of cancer: of all cancer cells, all the time?
Trends Mol Med. 18, 509-15 (2012).
Gene expression profiling associated with the progression to poorly differentiated thyroid carcinomas.
Br J Cancer. 101,1782-91 (2009).
Cell cycle deregulation and TP53 and RAS mutations are major events in poorly differentiated and undifferentiated thyroid carcinomas.
J Clin Endocrinol Metab. 99, E497-507 (2014).